By Alexandra Kent
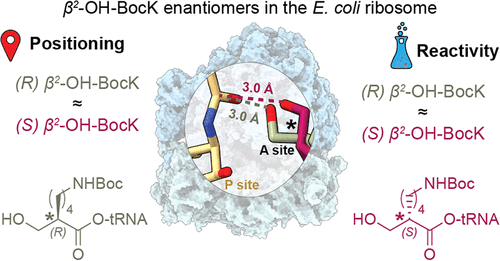
The goal of C-GEM is to generate ribosomally synthesized, sequence-defined polymers using a diverse set of non-natural monomers. Although ribosomes are known to incorporate some unnatural monomers, the basis for observed differences in incorporation efficiencies of different stereoisomers remains poorly understood. In this paper, C-GEM researchers examine two enantiomers of a β2-hydroxy acid that have dramatically different incorporation profiles in vivo. They demonstrate using cryo-EM that these stereoisomers are both positioned in a reactive conformation within the ribosome and show that both are incorporated with equal efficiency into peptides in vitro. This work demonstrates the tolerance that E. coli ribosomes have for monomers with modified backbones, and will aid in design of new monomers that are positioned optimally for reactivity.
This work involved contributions from the Cate lab and Schepartz lab.